NOVASIGN STUDIO | CDMO
Novasign Studio for CDMOs
Start with one question. Build a workflow. Keep it for the next project.
THE CHALLENGE
The overhead every high-performing CDMO team carries
Scale-up and tech transfer decisions are made against standardized workflows that do not always hold up when results need to be justified
Sponsors expect more than internal reasoning: process decisions need to hold up externally, and building that justification takes time that is rarely budgeted
The bottleneck is rarely expertise. It is the overhead built into how process development has always worked.
HOW NOVASIGN STUDIO HELPS
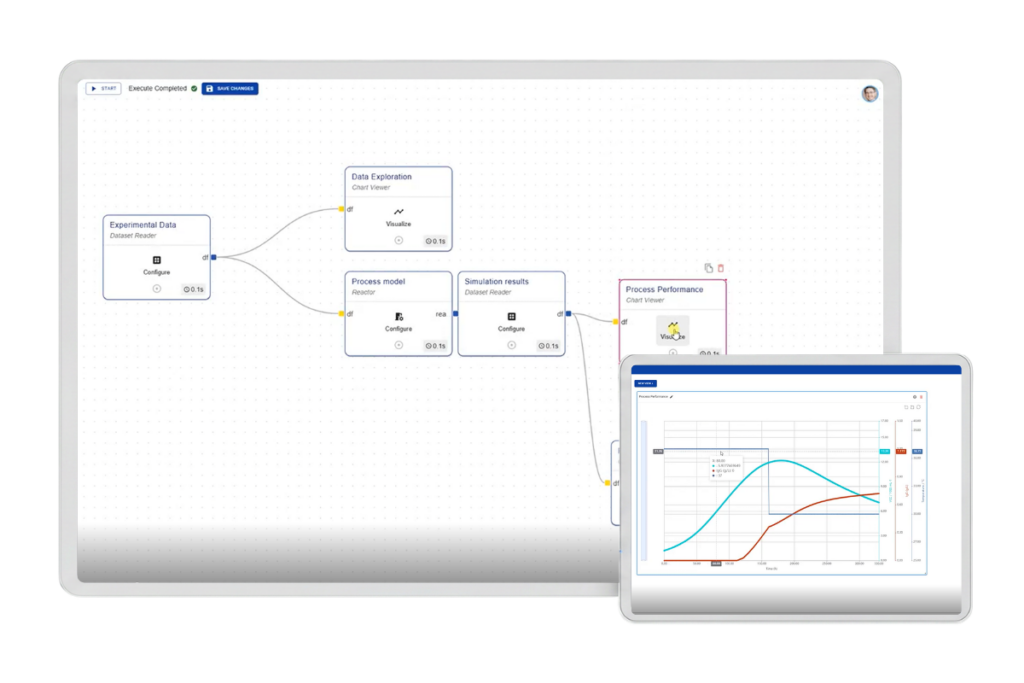
One connected workflow. From process data to well-justified decisions
Novasign Studio introduces a structured workflow layer across your development process.
Planning → Data processing → Process modeling → Decision support
Novasign Studio gives each step a structured home, open to extension where you need it:
1
Plan
Design smarter experiments around the specific decision you need to make, from screening to model-based Design of Experiments.
2
Process data
Consolidate campaign data from instruments such as bioreactors and PAT sensors, and from data systems such as LIMS and historian, into reproducible, comparable datasets your whole team can work from.
3
Model
Build process models from your data and process knowledge, explainable, auditable, and tailored to the program at hand.
4
Simulate
Run in-silico scenarios to test operating strategies, de-risk scale-up, and evaluate trade-offs before committing to lab or engineering runs.
5
Decide and reuse
Generate clear, explainable outputs that can be shared with clients, applied across programs, and built on over time, not rebuilt from scratch.
USE CASES: WHERE STUDIO FITS IN CDMO DEVELOPMENT
Where teams see the fastest return on time and resources
The best starting points are where your team already feels the overhead: repeated effort, inconsistent data, or uncertainty in scale-up decisions, across the entire bioprocess development chain.
Faster speed to clinic
Build a workflow around your platform process to reduce the experimental effort required for early development.
Process optimization and characterization can easily consume more runs than necessary when experiments are planned without a clear model to guide them. Novasign replaces this with model-guided experimental planning that is tailored to your specific process question and refined as data comes in.
Scale-up and tech transfer prediction
Scale-up is where CDMOs can demonstrate the strength of their process development strategy. Novasign integrates CFD-derived parameters such as kLa, mixing time, shear rate, and P/V directly into process models, enabling in-silico prediction of process performance at any target scale before running a single engineering run.
This adds a quantified, model-based layer on top of existing engineering rules and experience, rather than replacing them.
Single unit operations or the full bioprocess chain
Process knowledge that travels with the team
Valuable process knowledge is currently buried in spreadsheets, local scripts, and individual expertise. Novasign Studio gives every team member a structured way to build on what a colleague has already developed, so expert knowledge is not locked to one person but shared, reused, and extended across programs.
Deviation Investigation and CAPA support
When deviations occur, the first challenge is having enough traceable information to understand what happened and why. Novasign Studio’s reproducible data processing workflows ensure that every transformation step is documented and repeatable.
Process teams and quality teams see not just the data, but how conclusions were drawn, which process parameters were considered, and how the process was characterized at the time of the deviation.
NOVASIGN STUDIO
Two ways Novasign Studio support CDMOs
Novasign Studio improves how your team works internally and how you present your capabilities to clients and sponsors.
Build efficiency and reuse across every program
Reduce manual data wrangling and inconsistent interpretation across upstream and downstream programs. Give process teams reusable workflow support without adding modeling overhead or specialist resource requirements. Each workflow built today becomes a template for the next program.
Stand out as a digitally capable partner
Differentiate yourself from classic asset-driven CDMOs. Novasign Studio gives you a structured digital layer that makes process decisions faster, more consistent, and easier to justify, without overhauling your existing infrastructure. The result is a tangible differentiator you can show sponsors, not just describe.
WHY NOVASIGN STUDIO
Why Novasign Studio makes a difference for CDMO teams
01
Up to 70% fewer experiments
Learn more
Our workflows reduce experimental burden by 35–70% in process development, demonstrated across CHO, HEK, E. coli, and ATMP platforms. Fewer experiments means faster programs and lower material cost per client engagement.
02
Scale-up prediction using CFD modeling bioreactor
Learn more
Predict process performance at any target scale, including bioreactor geometry, kLa, and mixing time, before running engineering or pilot runs. Build client trust by arriving at tech transfer with a model, not a guess.
03
Reusable workflow foundation
Learn more
Capture process knowledge in structured workflows that teams can apply across programs, sites, and development stages.
04
Transparent, auditable decision support
Learn more
Every model assumption, preprocessing step, and decision path is visible and explainable. This is what regulators expect and what clients need to justify.
05
End-to-end process continuity
Learn more
Connect upstream to downstream unit operations in one workflow environment, with clear visibility from raw data to final process recommendation.
SELECTED CASE STUDIES
Results from CDMOs and process development teams
22 → 8
Bespark*bio reduced experimental runs from 22 to 8 (64% reduction in experimental effort), achieving approximately €40k in material cost savings and a 12-week development acceleration.
50% +
Bisy GmbH reduced development timelines by more than 50% in an initial proof-of-concept project, using Novasign Studio to accelerate process development for new products.
How Novasign Studio compares
Novasign Studio is built for teams that need a practical, end-to-end workflow, covering everything from experimental data to modeling, simulation, and better process decisions.
Novasign Studio vs. internal scripts and local models
Move from person-dependent analyses to reusable, structured workflows your whole team can review, apply, and build on.
Novasign Studio vs. cloud-based bioprocess modeling platforms
Keep full flexibility in deployment, workflows, and model integration, including fully on-premises setups where data governance requires it.
Novasign Studio vs. standalone solutions
Use Novasign Studio as an integrated workflow platform and as a client-facing tool in sponsor interactions. Selected components can also be white-labeled for CDMOs who want to embed the capability under their own brand.
Novasign Studio vs. pure mechanistic or pure ML modeling tools
Supports mechanistic, data-driven, and hybrid models across multiple unit operations. Choose the modeling approach that fits your data availability, and connect unit operations so changes propagate through the full workflow.
HOW TO GET STARTED WITH NOVASIGN STUDIO
Novasign Studio is available in two ways, depending on whether you want to start with a proof of concept or go straight to a full license.
OPTION 1: NOVASIGN STUDIO + PROOF OF CONCEPT
The right starting point if you are evaluating digital process solutions, building the case internally, or want to see concrete results before committing time and resources. We work with you on a scoped proof of concept project using your own process data, so you see the value of Novasign Studio in your specific context, not on a generic example.
Start with a guided project on a real process question
1
Scoping Call
Define one area to improve (e.g., USP to DSP, early stage to late stage and scale-up, screening to characterization)
2
Feasibility Check
Assess whether your existing data can support predictive modeling
3
Proof of Concept
Proof of Concept: We model and simulate your process workflow in Novasign Studio together with you
4
Results & Workshop
Clear outputs, recommendations, and a defined path forward
OPTION 2: NOVASIGN STUDIO LICENSE
The right starting point if you want direct access to the platform and the freedom to work at your own pace. You get full access to all capabilities, onboarding support, and ongoing assistance from the Novasign team.
Get full access and explore at your own pace
1
Access Novasign Studio
Receive your license, configure the platform for your environment, and choose the right deployment option
2
Optional Feasibility Check
Assess whether your existing data is suitable for predictive modeling before diving in
3
Onboarding Workshop
Get started on Novasign-provided reference process datasets covering all relevant unit operations
4
Work independently
Explore and build your own workflows, with full support from the Novasign team whenever you need it
Frequently asked questions
What do I need to get started with Novasign?
Less than you might expect. We begin with a short scoping call to understand your process and goals, with no data required upfront. If you want to move into a proof of concept, structured or semi-structured process data is sufficient (CSV, historian exports, LIMS, XLSX). We assess data suitability together before any commitment. Most customers start with a retrospective study of existing process data from a single unit operation.
Can I run Novasign Studio on-premises?
Yes. Novasign Studio can be deployed fully on-premises, giving your team complete control over your data and infrastructure. Cloud-based deployment is also available. This flexibility is an important differentiator for CDMOs with strict data governance, sponsor confidentiality requirements, or internal IT policies.
Do I need a dedicated modeling or data science team?
No. Studio is designed for process scientists, lab scientists, and engineers as the primary users. All core features are accessible through no-code graphical interfaces, with no programming required. Teams with data science capability can also use Studio: custom Python code integrates directly, letting data scientists bring their own models and scripts into a shared Novasign Studio environment so colleagues can use them immediately, without any coding on their end.
Who owns the models and workflows I build?
You do. All models, data processing workflows, and derived insights you generate in Novasign Studio belong to your organization. Novasign retains ownership of the platform's native components only. Your data is never used by Novasign for any other purpose.
Can I share results with my clients inside Novasign Studio?
Yes. Studio can serve as an interactive presentation layer between your team and your clients, replacing static slides and spreadsheet exports with live, explorable process views. You can share specific experimental and modeling outputs with a sponsor directly inside Novasign Studio, where they can interact with and continue working with the data. This gives clients a more transparent, engaging experience and positions your CDMO as a digitally capable partner.
Can my clients see each other's data?
No. The permission system is built to ensure strict data separation between clients. Each sponsor only ever sees the data, models, and workflows that belong to their own program. Access is controlled at every level, covering company, site, department, and user, so client confidentiality is maintained by design.
How many experiments do I need?
There is no fixed number, and that is precisely the point. Novasign Studio works with whatever data you already have, however limited, and uses it to reduce risk and identify the smallest set of experiments needed to reach your specific process goal. Rather than defining a standard DoE upfront, Novasign Studio builds on your existing process knowledge, trains a model, and identifies which experiments will be most informative next. The result is a targeted, goal-driven experimental plan, not a predefined one.
How quickly will I see results?
The first tangible results, including structured data, comparable datasets, and initial visualizations, are typically delivered within 1–4 weeks. Predictive models and reduction in experimental effort follow within 1–3 months, depending on data availability and project scope. Customers starting with a proof of concept typically see clear, decision-relevant outputs within the first project.
Can I start modeling before my data infrastructure is fully set up?
Yes. You do not need centralized data, a standardized data infrastructure, or any automation system before getting value from modeling. Most teams start by using models to support process decisions, reducing uncertainty and identifying the most informative next experiment before committing to a run. Automation is a natural next step, and when the time comes, models built in Novasign Studio can feed directly into automated workflows. In our experience, teams want to investigate and understand their process first, and automate once they trust the logic.
How transparent is Novasign Studio?
Fully transparent. Some software tools deliver results you have to trust without being able to investigate how they were derived; Novasign Studio is designed to be the opposite. Every data processing step, model training decision, simulation, and process recommendation is traceable and explainable. Your team can inspect and document the complete path from raw data to output, which is essential when justifying decisions to sponsors or supporting regulatory submissions.
GETING STARTED
Build digital capability.
One workflow at a time
Start with one process question that matters to your team.
Deliver a clear result, build confidence, and expand from there.