Next‑Generation Media Development: How Modeling Reduces Cost and Accelerates Bioprocess Optimization

Media composition is one of the least transparent elements in modern cell culture bioprocess development. Whether producing monoclonal antibodies, recombinant proteins, industrial enzymes, or cultivated food products, the composition of the cell culture or fermentation media directly determines growth, productivity, and product quality. Yet despite its importance, media development is still largely driven by empirical experimentation and incomplete process understanding.
For biotechnology companies, contract development and manufacturing organizations (CDMOs), and emerging sectors such as precision fermentation and cultured food, inefficient media optimization results in unnecessary development time, high experimental costs, and limited understanding of cellular metabolism.
Why Media Development Is Still a Major Bottleneck
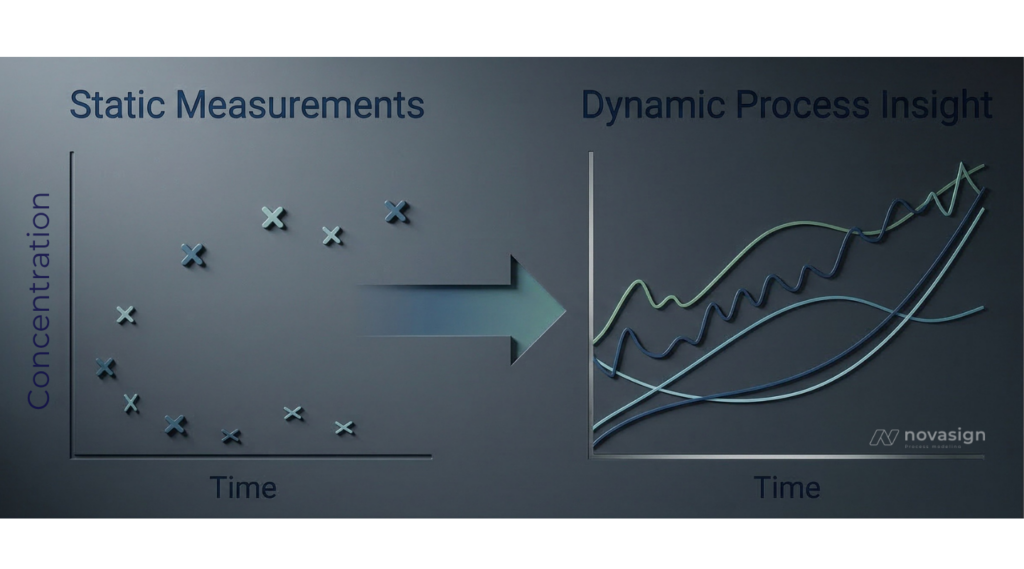
Traditional media development often relies on Design‑of‑Experiment (DoE) strategies that evaluate the impact of media components based on endpoint measurements. While still informative, these methods typically capture only static snapshots of the process and fail to reveal the dynamic relationship between nutrient availability, cellular uptake, and metabolic response.
Media Cost as a Barrier for Cultured Food
In emerging industries such as cultivated meat and precision fermentation, media cost has become one of the largest economic barriers to commercialization. Growth media for mammalian cell culture often contain expensive components such as amino acids, growth factors, vitamins, and recombinant proteins. In many cases, the cost of media alone dominates the overall cost of production.
Model‑guided media development enables companies to systematically identify which components drive productivity and which components can be reduced without affecting performance, creating a pathway toward economically viable large‑scale production.
Media Transparency Challenges in Biopharma
In biopharmaceutical manufacturing, many companies rely on commercially available off‑the‑shelf media formulations. While convenient, these formulations often function as black boxes: the exact concentration ranges, interactions, and limiting components remain unclear.
Many organizations are therefore investing in internal media development to understand how amino acids, carbon sources, vitamins, and growth factors influence cellular metabolism and recombinant protein production.
The Novasign Approach
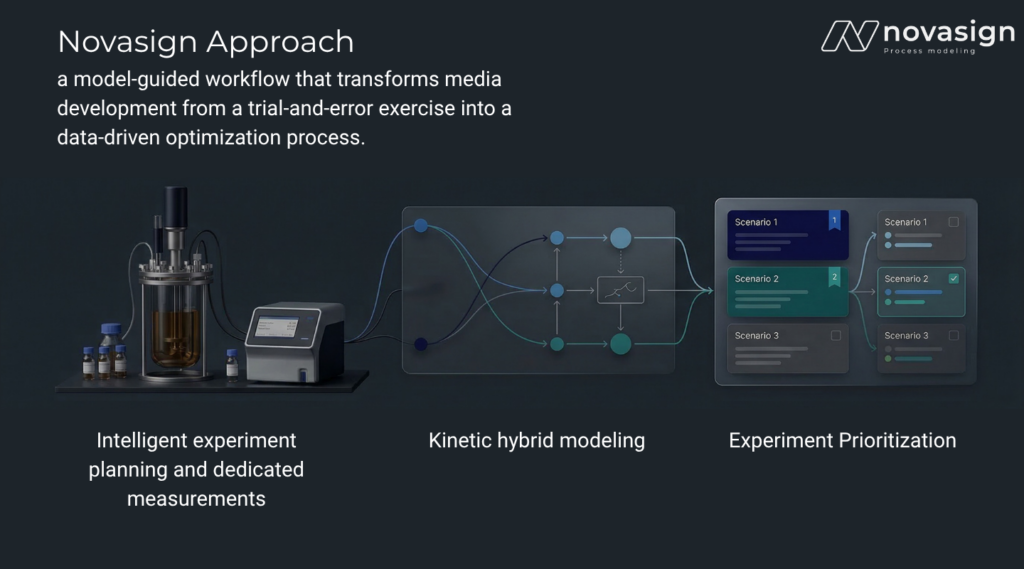
Novasign has developed a modeling‑guided workflow that transforms media development from a trial‑and‑error exercise into a data‑driven optimization process. The workflow uses intelligent experiment planning, dedicated measurements, and kinetic hybrid modeling to identify the media component concentrations with the greatest impact on process performance.
Instead of testing hundreds of combinations blindly, the Novasign platform identifies which components most strongly influence cellular performance and recommends how they should be dosed during the cultivation.
Case Study: 60% Fewer Experiments
In a recent industry collaboration, Novasign applied this workflow to optimize media and feed strategies. The model identified limiting amino acids and suggested optimized feeding concentrations. The approach reduced experimental effort by approximately 60% compared with classical DoE‑based media development while also increasing the titer of the final feeding strategy.
The Future of Media Development
By combining advanced analytics, metabolic modeling, and intelligent experiment design, Novasign enables companies to move beyond black-box media optimization and toward a predictive understanding of cellular nutrition.
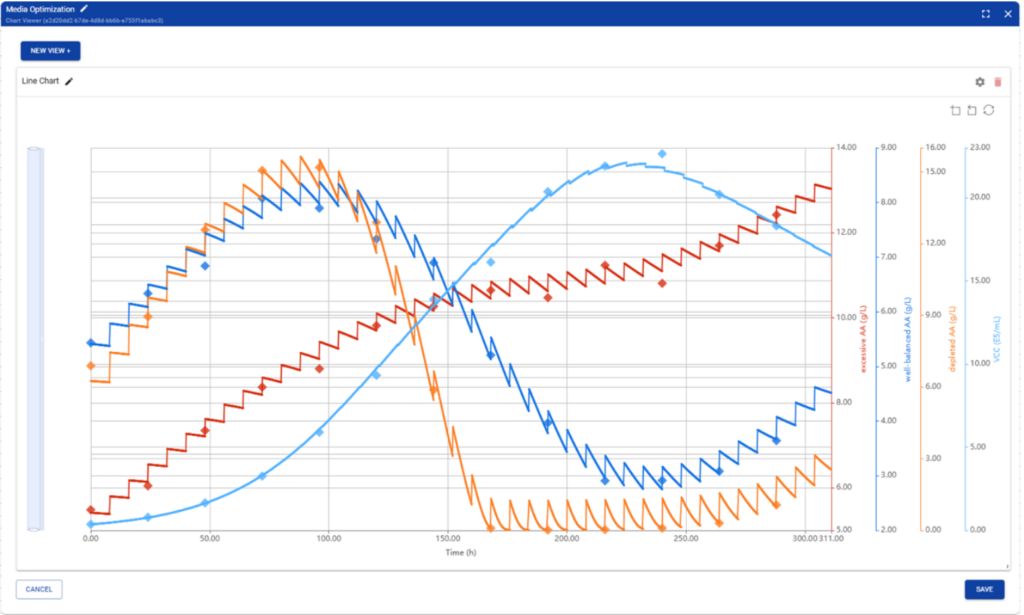
A key component enabling this shift is the ability to measure individual nutrients during cultivation. Analytical technologies such as HPLC or mass spectrometry allow researchers to quantify amino acids, carbon sources, and other media components over time, providing valuable insight into nutrient consumption and metabolic dynamics.
For faster and more practical implementation during process development, instruments such as the REBEL XT from Repligen enable rapid amino acid analysis directly during cell culture experiments. The system allows process developers to monitor amino acid depletion and accumulation during cultivation, making it possible to identify limiting nutrients much earlier in the development process.
When these measurements are integrated into the Novasign modeling workflow, they provide a powerful feedback loop: experimental data reveals which nutrients become limiting, while the model predicts optimal feeding concentrations that maximize productivity without exceeding solubility limits or causing nutrient accumulation.
Together, advanced analytics and predictive modeling create a new paradigm for media development – one that not only reduces experimental workload but also provides a deeper mechanistic understanding of how individual media components influence cellular behavior and process performance.
Optimize media faster with fewer experiments.
Discover how Novasign Studio helps teams identify the right media and feeding strategy with up to 60% less experimental effort.