De‑Risking Bioprocess Scale‑Up: Predictive Modeling for Reliable Fermentation and Cell Culture Manufacturing

Scaling a bioprocess from laboratory development to industrial manufacturing is one of the most critical, and risky steps in biotechnology. For contract development and manufacturing organizations (CDMOs), large pharmaceutical companies, industrial biotechnology producers, and emerging sectors such as cultured food and precision fermentation, scale‑up failures translate directly into lost time, wasted resources, and delayed commercialization or in worst case end of business.
Why Bioprocess Scale‑Up Often Fails
Bioprocess development typically starts in small reactors ranging from a few milliliters to a few liters. However, commercial production often takes place in reactors that are thousands of times larger. During this transition, cells experience very different environmental conditions due to changes in mixing, oxygen transfer, nutrient gradients, and hydrodynamic forces.
Even when key engineering parameters appear identical, the cellular environment in a production‑scale bioreactor can differ dramatically from the laboratory setting. These differences frequently lead to unexpected changes in cell growth, metabolism, product formation, or product quality.
The Need for Predictive Scale‑Up in Biomanufacturing
Traditional scale‑up strategies rely on simplified engineering correlations such as constant power input, tip speed, or oxygen transfer coefficients. While useful, these approaches do not fully capture the complex interaction between cellular metabolism and reactor physics.
As a result, scale‑up often becomes an iterative trial‑and‑error process that requires multiple expensive engineering runs. Each failed or sub‑optimal run increases development time, consumes budget, and delays commercialization.
A New Approach: Integrating Cellular Models with Reactor Physics
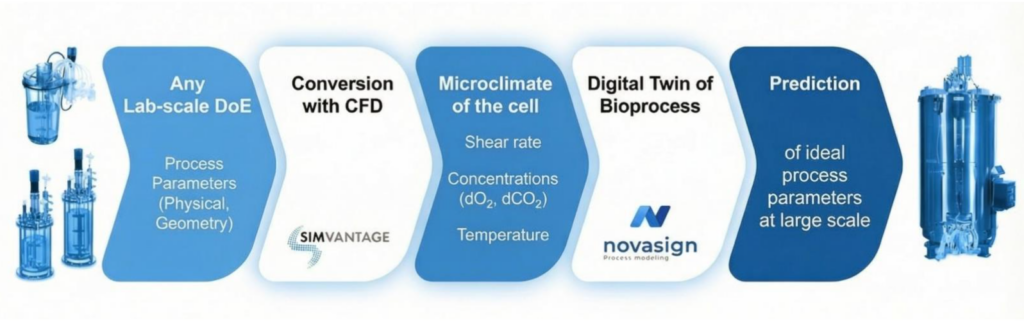
During the 5 years of collaboration Novasign and SimVantage have developed an integrated modeling framework that combines biological process modeling with rapid‑response reactor simulations. The goal is simple: predict how cells behave in industrial bioreactors before the first large‑scale experiment is performed.

Training the Cellular Model at Small Scale
The Novasign platform creates data‑driven models describing cellular metabolism and physiological responses. These models are trained using small‑scale experiments conducted under both ideal and deliberately sub‑optimal conditions. This approach captures how cells react to environmental stresses such as nutrient gradients or oxygen limitations.
Capturing the Large‑Scale Environment
SimVantage complements this approach by modeling the physical environment of both the small-scale and industrial bioreactors. The resulting rapid‑response models simulate mixing behavior, mass transfer, and spatial gradients that cells experience during the cultivation.
This integrated framework effectively creates a predictive digital twin of the bioprocess. It allows process developers to evaluate scale‑up strategies, optimize operating conditions, and identify potential limitations before performing large‑scale experiments.
Case Study: Predictive Scale‑Up of Amino Acid Fermentation
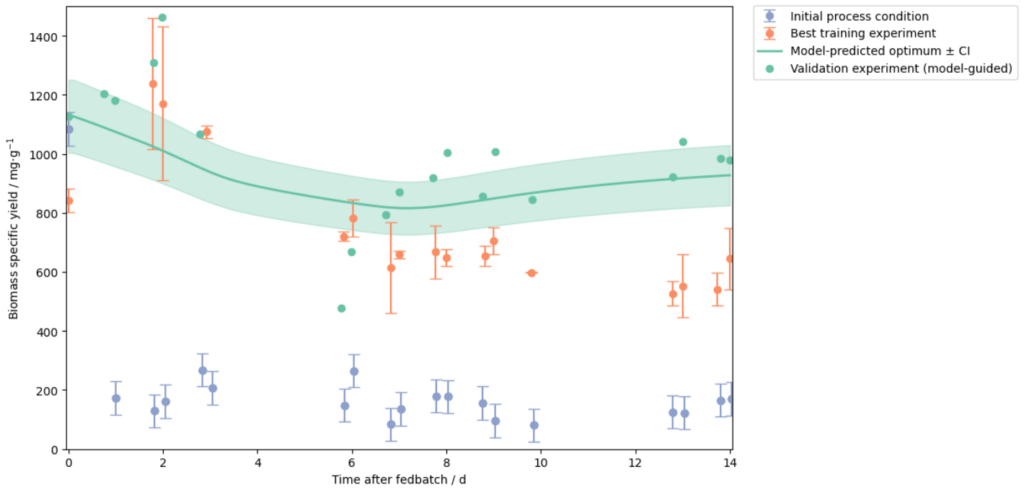
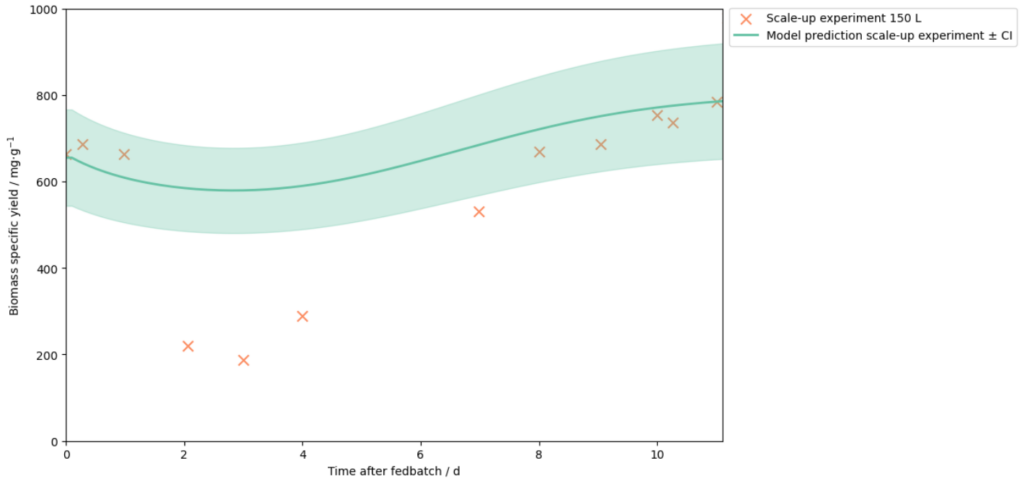
A recent case study demonstrates the power of this approach. Novasign optimized an amino acid fermentation process in lab-scale and then used the integrated Novasign–SimVantage modeling framework to predict its behavior in a 150 L continuous bioreactor.
By training the cellular model on experimental observations and combining it with the reactor physics model, the system predicted the optimal large‑scale process outcome before the experiment was performed.
Benefits for CDMOs, Biopharma, and Industrial Biotechnology
For CDMOs, predictive scale‑up significantly reduces the number of engineering runs required for process transfer and improves reliability during client technology transfers.
For pharmaceutical manufacturers, the approach improves confidence in CMC development and reduces the risk of unexpected process deviations during clinical or commercial manufacturing.
Industrial biotechnology companies and precision fermentation startups benefit from faster commercialization and lower capital risk when designing large‑scale fermentation infrastructure.
The Future of Bioprocess Development
Predictive modeling is transforming how bioprocesses are developed and scaled. By integrating biological insight with reactor‑scale physics, companies can move from empirical scale‑up toward a data‑driven and predictive development paradigm.
With the combined Novasign and SimVantage approach, developers can understand how cells behave at production scale long before the first large‑scale experiment takes place, dramatically reducing risk and accelerating the path from laboratory discovery to industrial manufacturing.
De-risk scale-up before you run it.
Use predictive modeling to understand large-scale performance earlier and reduce costly surprises during tech transfer.